Atriva Therapeutics has been working to develop flu treatment for several years. Now the company is working on a medicine to help people suffering from COVID-19.

The team from Atriva Therapeutics develops medicines to combat influenza viruses. The new medicine looks as if it could help those suffering from coronavirus.
In July ATR-002 will take its next step. It could be the penultimate one. At the end of the final stage – if all trials are successful – ATR-002 would become an approved medicine. It could be used to treat people with severe COVID-19.
As early as mid-February, Rainer Lichtenberger suspected that a pandemic was spreading, because the situation already resembled crisis scenarios. And because biotechnology company Atriva Therapeutics, of which Lichtenberger is the CEO, has good scientific contacts in China. “It was clear that it would be at least as bad as the worst flu,” says Lichtenberger.
Atriva has been working on influenza viruses since it was founded in 2015. Scientists at the start-up, a spin-off from Tübingen, Giessen and Münster universities, developed candidate drug believing that it could be used to combat severe influenza and other serious respiratory diseases caused by viruses.
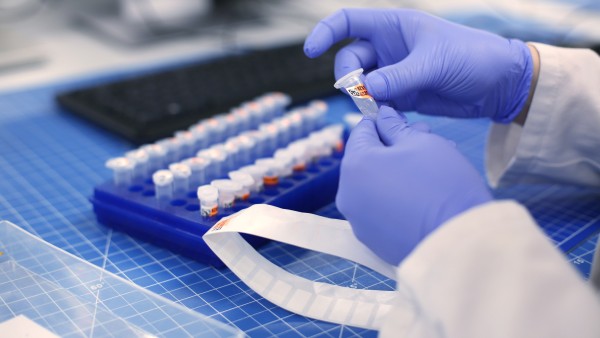
The years of preparatory work are now enabling Atriva, during the COVID-19 pandemic, to take its product straight into the second of a probable three clinical trial phases. . For the month of July, Atriva is planning the start of a multinational study under the guidance of Charité Berlin. It is envisaged to treat moderately severe to severe COVID-19 cases with ATR-002. The German Federal Institute for Drugs and Medical Devices showed high interest and has already been very helpful during scientific consultations. For developing and producing the test medication, Atriva is working with its long-standing partner Evonik, a German specialty chemicals company.
Start-up funding by KfW Capital
Alongside the work of the scientists, Lichtenberger looks after financing. Atriva needs several million euros. ”We need to go for it now,“ says the CEO, who is also hoping to attract public funding. He estimates that around 60 million euros will be needed to bring the new drug to market to treat the important indications of influenza, and in the future also of COVID-19.
High-Tech Gründerfonds (HTGF) was one of the start-up's initial investors, having come on board in 2016. In turn, one of HTGF's main investors is KfW Capital, the investment subsidiary of KfW Group. “The current coronavirus crisis shows how important it is to finance alternative therapeutic approaches which don't draw the attention of the pharmaceutical industry and venture capital firms. High-Tech Gründerfonds made a major contribution to this during Atriva's “seed phase” and we hope that in doing so, we helped to drive the development of a wide-ranging therapy to treat viruses like coronavirus or influenza,” says Frank Hensel, Principal of HTGF.
Alexander Thees, managing director of KfW Capital adds: “The example of Atriva shows how important it is for new ideas for fighting viral respiratory diseases to find suitable financing at an early stage – especially since the road to the final product in the life sciences is often long and rocky. KfW Capital has been investing in the High Tech Start-up Fund since the first generation of funds; it focuses on very young technology companies and thus contributes to turning ideas into marketable products, in the case of Atriva potentially life-saving drugs."
How the drug works
The developed candidate drug inhibits a signaling cascade in the host cells, for instance in the lungs, which the virus needs in order to reproduce. In addition, ATR-002 influences the immune response in a way that helps the body to fight the life-threatening symptoms of severe COVID-19 cases. Atriva has applied for the corresponding patents at the European Patent Office. According to Lichtenberger, there are three points which make COVID-19 more problematic than flu: there is no vaccine, there are no medicines and the incubation period is much longer.
We do not yet know how many victims the coronavirus pandemic will claim. However, we do know how many die during the annual flu outbreak: influenza puts between five and ten million people in the high-risk groups in hospital each year. In bad years, over half a million of these people will die of it. With 360,000 deaths , the COVID-19 pandemic unfortunately has already almost reached these figures, and there is no end in sight!
“We are looking at large sections of the population,” says Lichtenberger of his company's objectives, “and we have a clearly-structured development plan”. The pharmacist has been working for life science companies for 30 years, and has spent half of this time leading them. He was thus all the more perplexed about “how hard it is for Atriva to find large investors”. Many venture capitalists prefer the field of oncology because with cancer there are “large and predictable market developments”. In contrast, in Lichtenberger's words, research into infectious diseases has been “criminally neglected for a long time”.

“Research into infectious diseases has been criminally neglected for a long time.”
It is tragic that it took a catastrophe to trigger a rethink, “but the wind is now changing”. Sars-CoV-2 will no longer be a significant danger once herd immunity has been established. It is just that no-one knows when this will happen. But Atriva continues to work on its candidate. The drug is due to be tested on patients in late 2020 during the next flu season. For this indication too, there are no longer any scientific and regulatory barriers to its use in clinical phase II.
When it comes to dealing with the coronavirus pandemic, Lichtenberger feels that he is in good hands in Germany. In his view, the crisis is being managed well. “We are doing better than any other large European country,” he even states.
The described project contributes to the following United Nationsʼ Sustainable Development Goals
Goal 3: Ensure healthy lives and promote well-being for all at all ages
Health is the goal, prerequisite and result of sustainable development. Supporting health is a humanitarian requirement – both in developed and developing countries. Around 39 per cent of the worldʼs population lives without health insurance. In poor countries, this amount even exceeds 90 per cent. Many people still die from diseases that are not necessarily fatal with the right treatment, or that could easily be prevented with vaccinations. Strengthening health systems, particularly by making vaccines widely available, can make it possible for us to drive these diseases back and even eradicate them by 2030.

All United Nations member states adopted the 2030 Agenda in 2015. At its heart is a list of 17 goals for sustainable development, known as the Sustainable Development Goals (SDGs). Our world should become a place where people are able to live in peace with each other in ways that are ecologically compatible, socially just, and economically effective.
Published on KfW Stories: 30 April 2020.












Data protection principles
If you click on one of the following icons, your data will be sent to the corresponding social network.
Privacy information